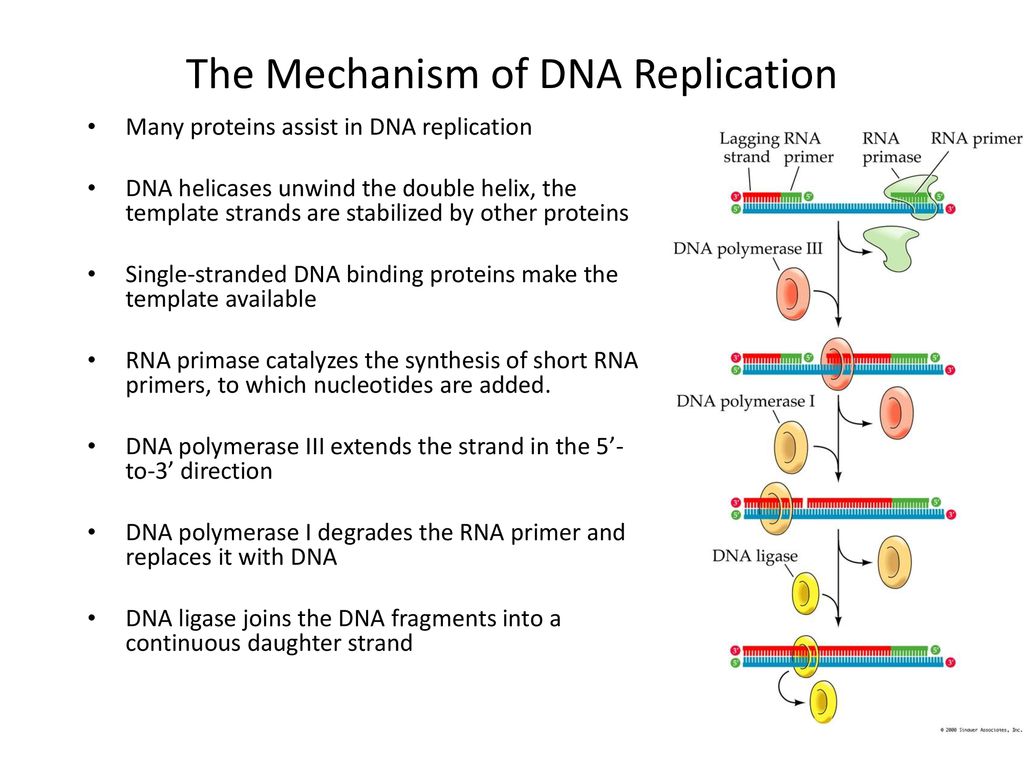
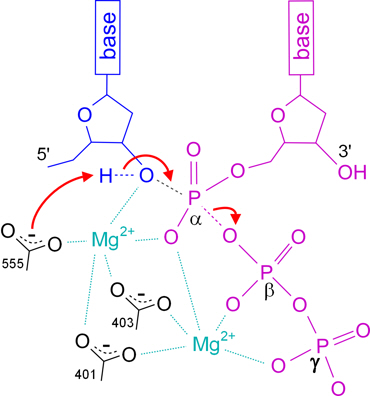
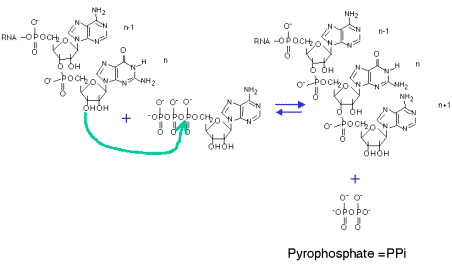
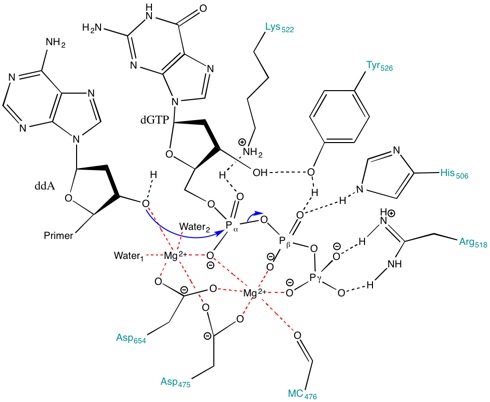
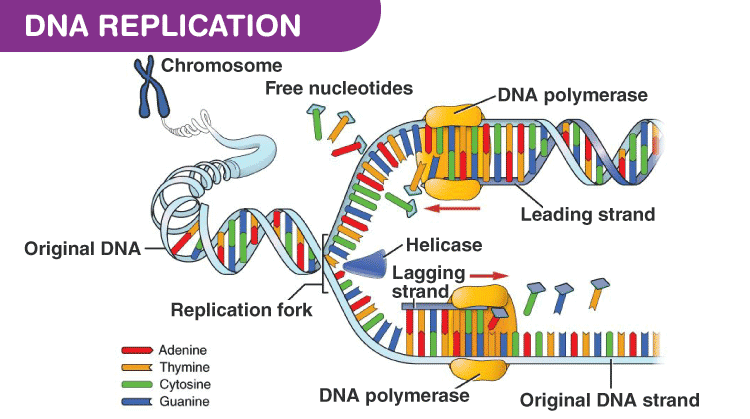
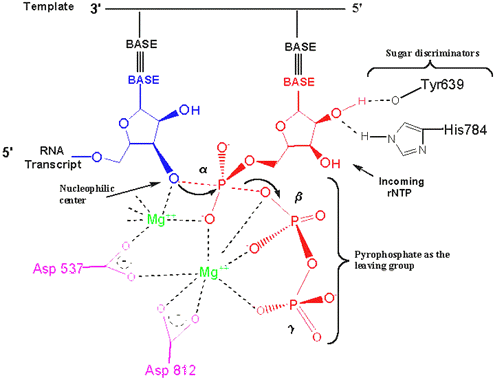
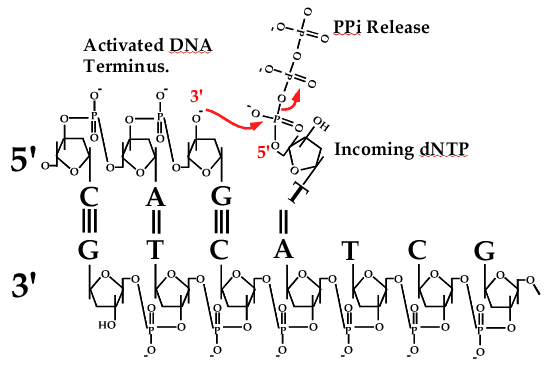
Catalytic mechanism of DNA polymerases—Two metal ions or three? - Tsai - 2019 - Protein Science - Wiley Online Library

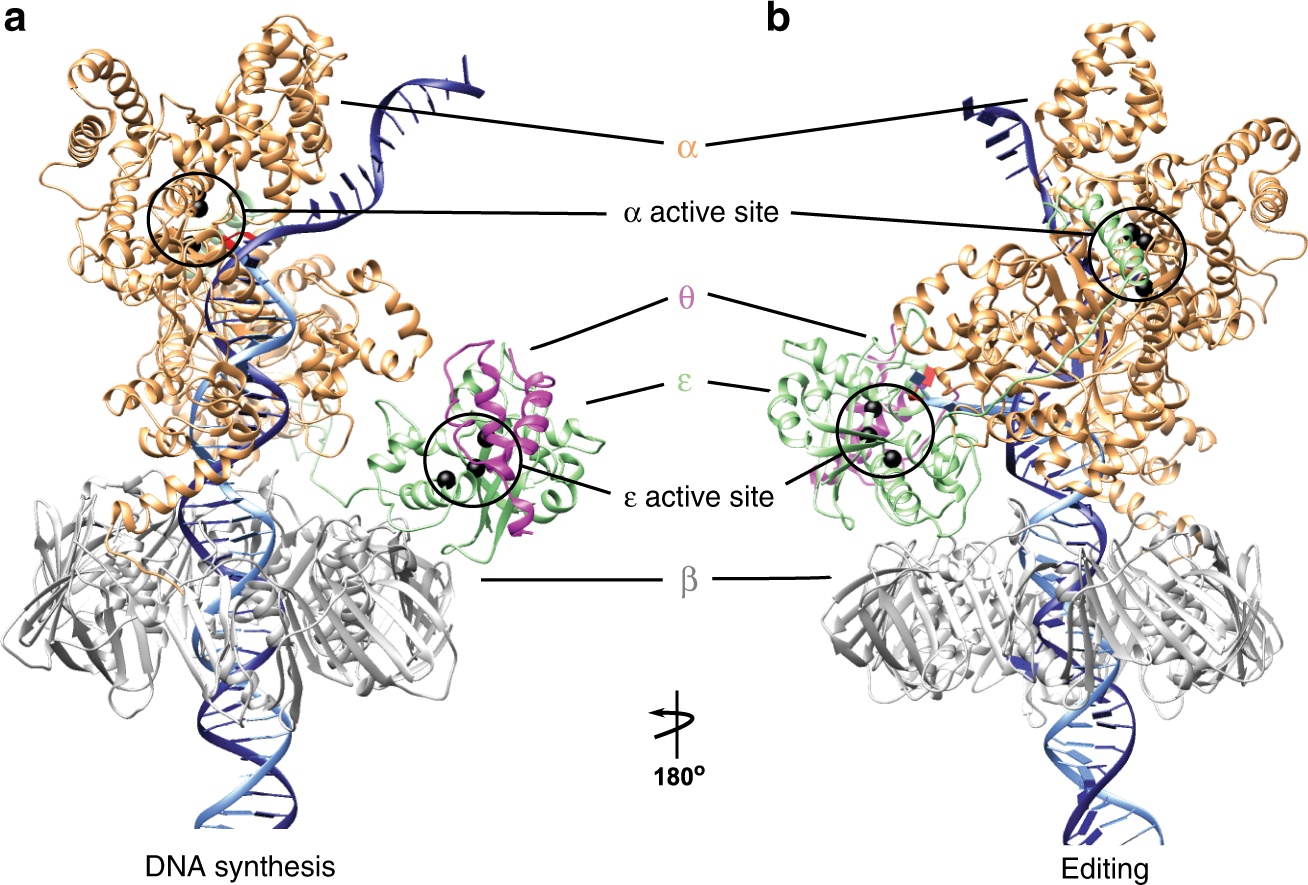
Reaction mechanism of the epsilon subunit of E. coli DNA polymerase III: insights into active site metal coordination and catalytically significant residues. | Semantic Scholar

A fidelity mechanism in DNA polymerase lambda promotes error‐free bypass of 8‐oxo‐dG | The EMBO Journal

Mutational and pH Studies of the 3′ → 5′ Exonuclease Activity of Bacteriophage T4 DNA Polymerase* - Journal of Biological Chemistry
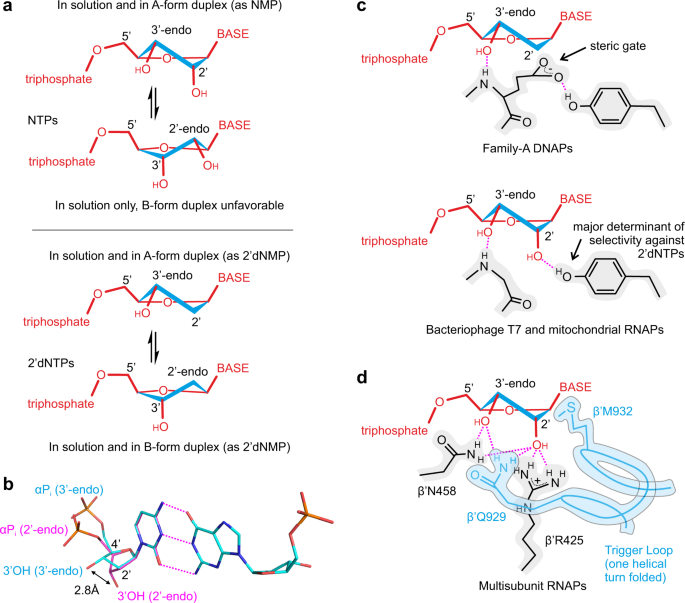
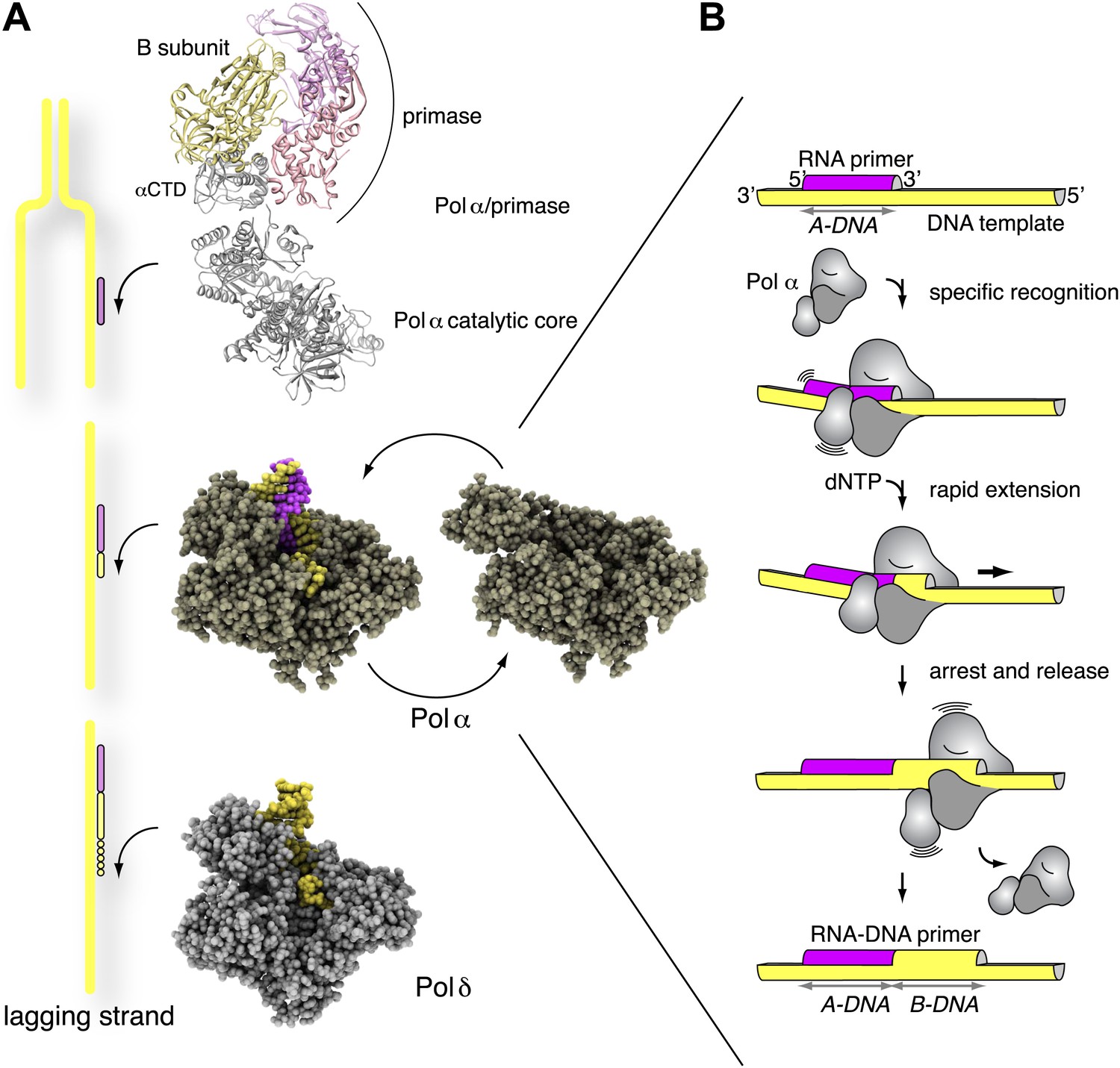
The mechanism of the nucleo-sugar selection by multi-subunit RNA polymerases | Nature Communications

Mechanism of incorporation of dTTP opposite dA by DNA polymerase IV.... | Download Scientific Diagram

Critical Role of Magnesium Ions in DNA Polymerase β's Closing and Active Site Assembly | Journal of the American Chemical Society
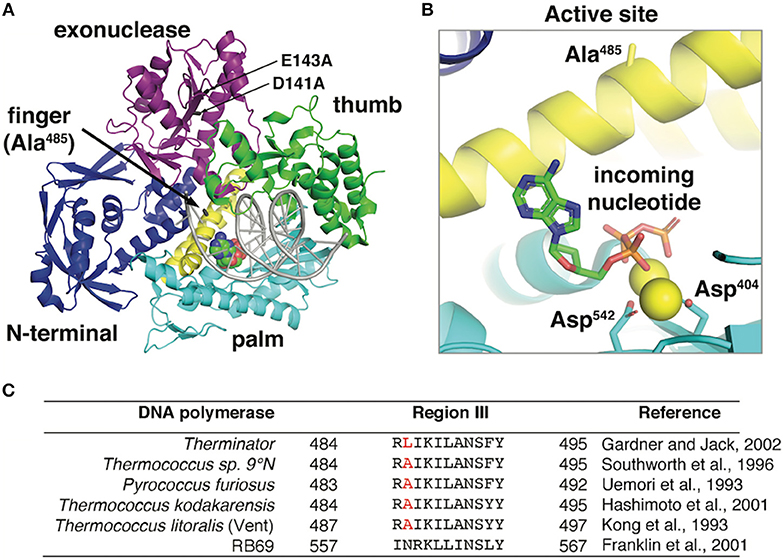
Frontiers | Therminator DNA Polymerase: Modified Nucleotides and Unnatural Substrates | Molecular Biosciences

Proposed exonuclease catalytic mechanism for T7 DNA polymerase. The... | Download Scientific Diagram